|
Different factors must be taken into account in the energy expression, including the interface between the particle/oil and particle/water phases. 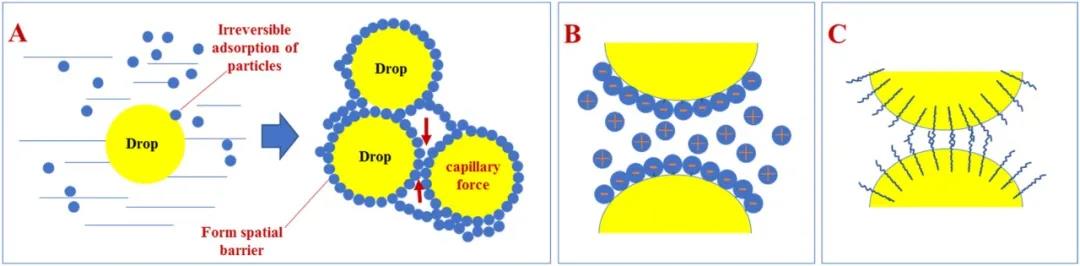
This is one of the driving forces contributing to particle transfer at the interface. Their adsorption leads to a decrease of the surface of the high-energy oil–water interface. 
Solid particles can be adsorbed at the interface between oil and water only upon partial wetting by both phases (i.e., dual wettability). In Pickering emulsions, the absorption of solid particles at the interface between liquids should form an obstacle to limit the merging (coalescence) between droplets. In classical emulsions, the system stability is ensured by the adsorption of amphiphilic compounds that modify the interfacial properties of the two phases. In these colloidal suspensions, stabilization is achieved by using only solid particles (hence the alternative name of solidstabilized emulsions) in the place of organic surfactants and polymers. Ramsden and Pickering were the first to identify and describe Pickering emulsions at the beginning of the 20th century. Here, we review recent findings concerning Pickering emulsions, with a particular focus on how the nanoparticles morphology (i.e., cube, ellipsoid, nanosheet, sphere, cylinder, rod, peanut) influences the type and stability of such emulsions, and their current applications in different fields such as antibacterial activity, protein recognition, catalysis, photocatalysis, and water purification. Moreover, the nanoparticle morphology strongly influences Pickering emulsion stability as well as the potential utilization of such emulsions. Therefore, they can be considered attractive components for various uses, such as photocatalysis and the preparation of new materials. Furthermore, Pickering emulsions show higher stability, lower toxicity, and stimuli-responsiveness, compared with emulsions that are stabilized by surfactants. In contrast to classical emulsions, in Pickering emulsions, solid microparticles or nanoparticles that localize at the interface between liquids are used as stabilizers, instead of surfactants, to enhance the droplet lifetime. 
In recent years, Pickering emulsions and their applications have attracted a great deal of attention due to their special features, which include easy preparation and enhanced stability.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
